Shiga toxin-producing Escherichia coli (STEC) are significant zoonotic pathogens worldwide, causing diseases ranging from diarrhea to hemolytic uremic syndrome (HUS). Ruminants, mainly cattle, are considered the primary reservoirs; however, the role of horses in the epidemiology of STEC remains poorly understood in Nigeria. A total of 231 rectal swabs were collected from horses across selected equine facilities in five Nigerian states. Samples were processed following standard procedures, and isolates were confirmed using the Analytical Profile Index (API 20E) kit. Antibiotic susceptibility testing (AST) was performed using the Kirby-Bauer disk diffusion method with a panel of nine antibiotics, and the isolated Escherichia (E.) coli were classified as sensitive or resistant according to CLSI guidelines. The Multiple Antibiotic Resistance (MAR) index was calculated to assess the risk of contamination. The E. coli isolates were screened for selected Shiga toxin genes (stx1, stx2 and eae) using polymerase chain reaction (PCR). Fourteen isolates (6%) were identified as Shiga toxin-producing E. coli; of these, 78.6% (11/14) expressed at least one virulence gene: 42.9% (6/14) carried stx2, 35.7% (5/14) carried eae, and no isolate carried stx1. All confirmed STEC isolates exhibited multidrug resistance (MDR). The isolates showed complete resistance (100%) to cefuroxime and ciprofloxacin, while all were susceptible to ofloxacin. MAR indices ranged from 0.2 to 0.8, with 77.8% of isolates exhibiting MAR indices ≥0.5. This study reports the detection of MDR STEC in horses in Nigeria, revealing high-level resistance to clinically important antibiotics and significant carriage of virulence genes. The high MAR indices indicate high-risk contamination sources. The detection of MDR STEC in this study underscores a significant health risk to horses, with potential for zoonotic transmission to humans and the environment.
Not applicable
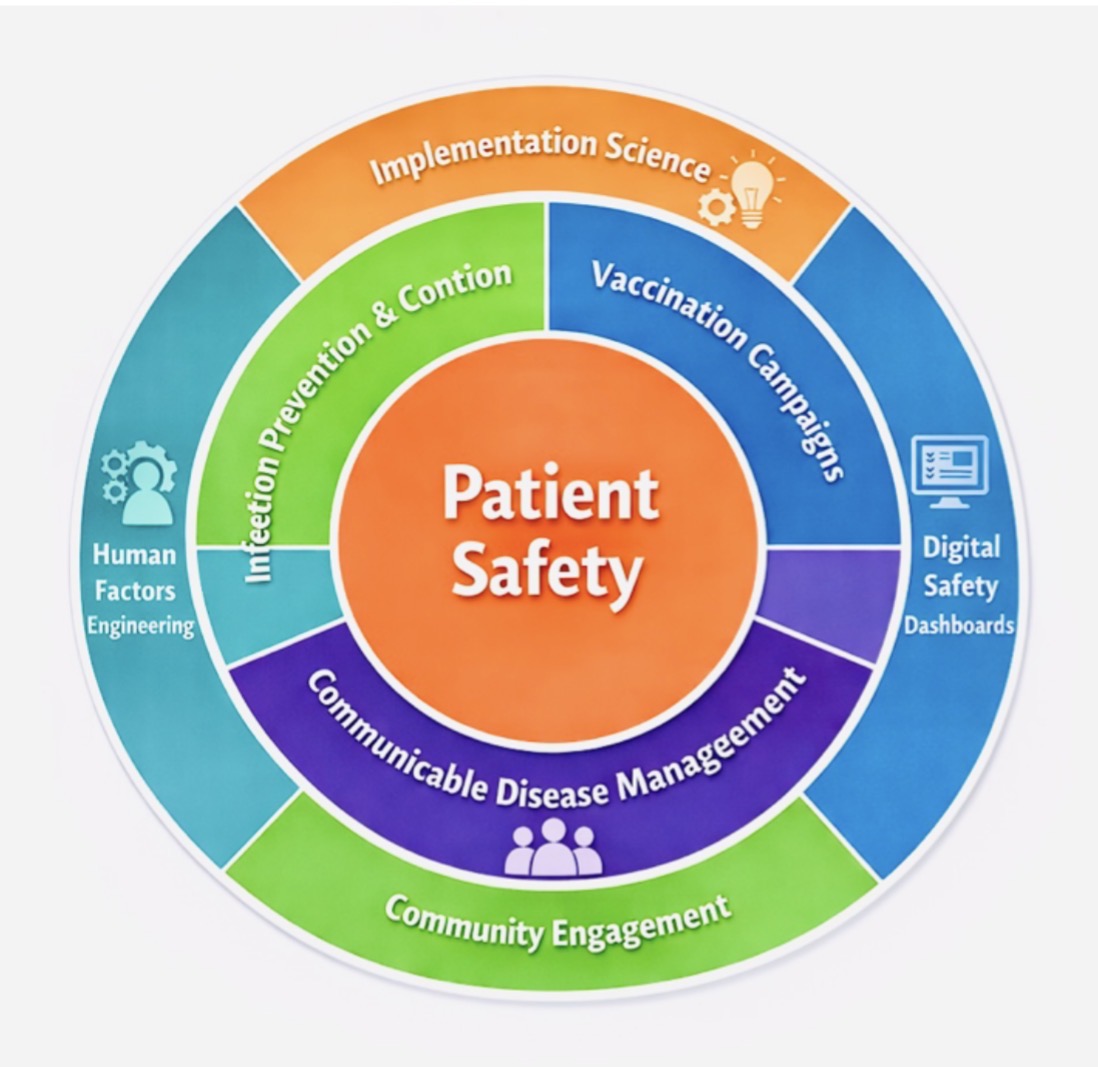
Patient safety is essential, and Infection Prevention and Control (IPC) has been shown to prevent harm. In primary care and outpatient settings, rising patient volumes, new care models, and emerging infectious threats require integrated safety systems. Aster Clinics in the United Arab Emirates (UAE) and Bahrain launched a comprehensive safety program that combined IPC, vaccination, and disease prevention strategies. The program was implemented from July 2025 to December 2025 across 80 clinics. Using a mixed-methods approach, key measures included standardized protocols, staff training, real-time audits, digital safety dashboards, vaccination outreach, and community campaigns. Quantitative data included IPC compliance, vaccination rates, and incident reports, while qualitative data assessed staff engagement, confidence, and safety culture. The program improved hand hygiene, vaccination rates, and transparency in incident reporting, fostering organizational learning. Digital tools enabled early detection and rapid response to safety issues. Staff gained confidence and participated more actively in safety discussions. Integrating IPC, vaccination, and disease management within a systems-based approach can enhance primary care safety, demonstrating how implementation science, human factors, digital tools, and community efforts strengthen resilient healthcare systems and promote a lasting safety culture.
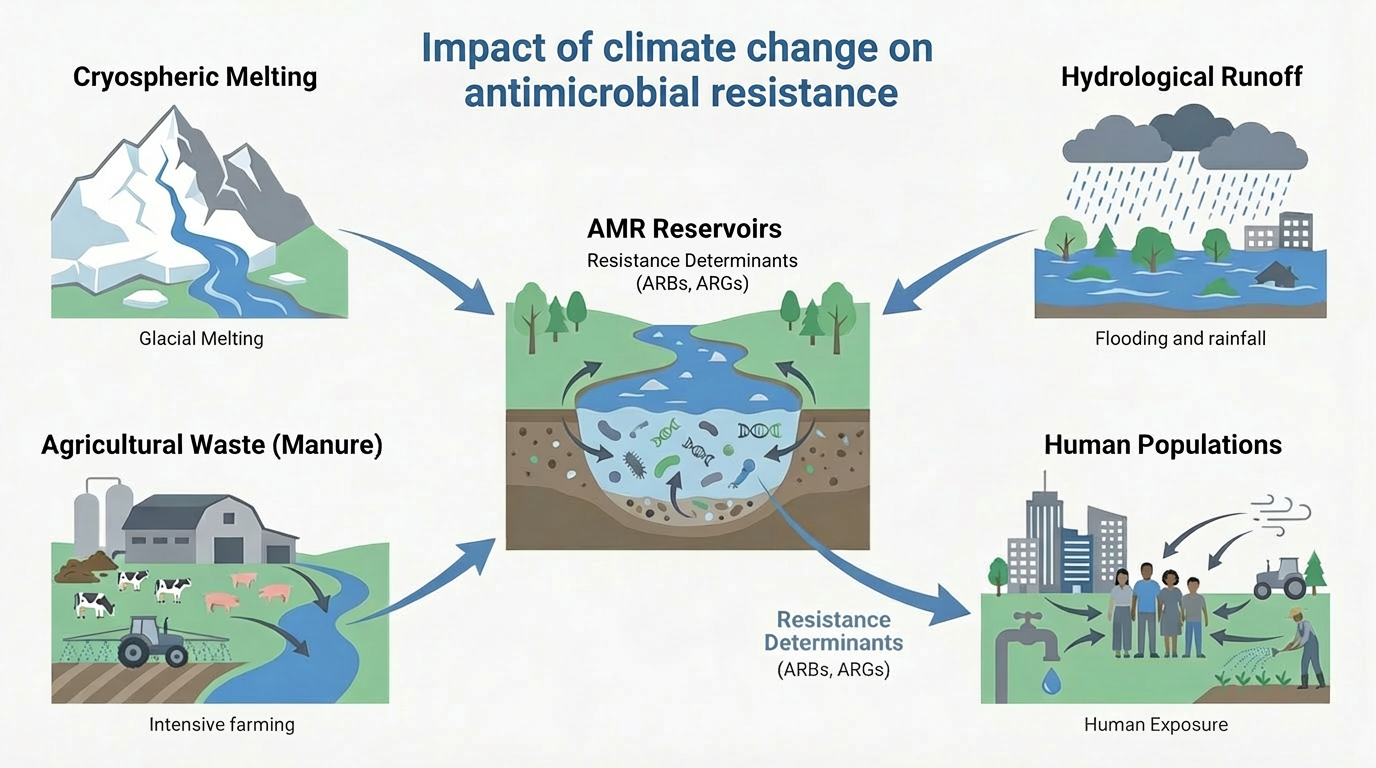
Antimicrobial resistance (AMR) and climate change are increasingly recognized as interconnected global threats driven by shared ecological, infrastructural, and socioeconomic vulnerabilities. This review summarizes emerging mechanistic and epidemiological evidence demonstrating how rising temperatures, hydrological extremes, ecosystem disruption, and environmental pollution accelerate the selection, persistence, and spread of antimicrobial resistance genes at the human-animal-environment interface. We emphasize climate-sensitive pathways, such as heat-driven microbial evolution, hydrological mobilization of resistance factors, aerosolized transmission, and climate-stressed food systems, which collectively reshape the distribution and impact of resistant bacterial infections. Importantly, we identify practical, co-beneficial interventions including pollution control, climate-resilient water and sanitation systems, adaptive antimicrobial stewardship, AI-powered surveillance, and integrated One Health governance. Framing AMR as a climate-sensitive health outcome shifts it from a mostly reactive, downstream clinical challenge to a predictable, preventable upstream systems vulnerability influenced by climate dynamics. Incorporating climate awareness into AMR policies and actions is therefore vital to preserving antimicrobial effectiveness, bolstering health system resilience, and safeguarding climate-sensitive populations in a rapidly warming world.